Презентация - Types of Chemical Reactions Classes of Chemical Compounds
Нужно больше вариантов? Смотреть похожие Нажмите для полного просмотра 
|
Распечатать
- Уникальность: 93%
- Слайдов: 38
- Просмотров: 5143
- Скачиваний: 2664
- Размер: 2.75 MB
- Онлайн: Да
- Формат: ppt / pptx
Слайды и текст этой онлайн презентации
Слайд 1

Types of
Chemical Reactions Classes of Chemical Compounds
Слайд 2

Topics Naming chemical compounds Revision (Periodic Law) Types of chemical reactions Classes of inorganic compounds and their properties
Слайд 3

Compounds substances composed of more than one element, chemically combined. A compound is represented by its chemical formula, a notation that uses atomic symbols with numerical subscripts to convey the relative proportion of atoms of different elements in the substance. E. g. HCl, H 2 O, NH 3 There are three fundamental kinds of chemical bonds between atoms - covalent bonds, ionic bonds and metallic bonds.
Слайд 4

Which of the following shows how the atomic radius of the elements changes on crossing from left to right in the row of the Periodic Table from potassium to bromine? K to Ca Sc to Zn Ga to Br A decrease increase decrease B decrease decrease increase C decrease decrease decrease D increase decrease increase E increase increase increase
Слайд 5

The atomic number of magnesium is 12. Which electron configuration given below corresponds to the Mg 2 ion (in its ground state)? A. Is 2 2s 2 2p 6 3s 2 3p 2 B. Is 2 2s 2 2p 6 3s 2 C. Is 2 2s 2 2p 6 D. Is 2 2s 2 2p 6 3s 1 E. Is 2 2s 2 2p 5 3s 1
Слайд 6

The alkali metals all react with water. a Describe what happens as each of lithium, sodium and potassium reacts with water. b State the difference in the reactivity of these alkali metals with water. с Describe what you could do experimentally to show what the product(s) are .
Слайд 7

Which one of the following is NOT the correct formula for a lithium compound? A Li 2 S B Li CO 3 C CH 3 CO 2 Li D Li HSO 4 E Li 3 N
Слайд 8

Organic and Inorganic Compounds
Chemical compounds can be classified as organic or inorganic. Organic compounds are those formed by carbon and hydrogen (hydrocarbon) or carbon and hydrogen together with oxygen, nitrogen, and a few other elements. Inorganic compounds are compounds composed of elements other than carbon. Except a few simple compounds of carbon, including carbon monoxide, carbon dioxide, carbonates and cyanides are generally considered to be inorganic.
Слайд 9

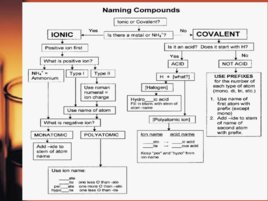
Naming of Chemical Compounds Chemical nomenclature is the system of names that chemists use to identify compounds. Two classes of names exist: common names and systematic names. Common names: ammonia, water, baking soda, laughing gas, muriatic acid, table salt Systematic names precisely identify the chemical composition of the compound. The present system of inorganic chemical nomenclature was devised by the International Union of Pure and Applied Chemistry (IUPAC).
Слайд 10
Слайд 11

Inorganic Compounds
Слайд 12

It s your turn Name the compounds SO 2 Fe(OH) 2 HCl HCl(aq) Cu Cl 2 , HNO 3 Cl 2 O 7 Ba SO 4 KNO 3 H 2 Si O 3 NH 4 Cl H 2 SO 4 Na HCO 3 (Cu OH) 2 CO 3 2. Write the formulas diphosphorus trioxide, iron dichloride, hydrogen sulfide, phosphoric acid, ammonia, sodium nitrite, phosphine, sulfurous acid, aluminium hydroxide, potassium dihydrocarbonate, sodium dichromate, sodium hexahydroxogermanate
Слайд 13

How many of the following compounds are acidic, alkaline or amphoteric (react with both acids and alkalis)? Al 2 O 3 Cl 2 O 7 CO 2 HCl H 3 PO 4 K 2 O KOH Mg O Na 2 O NO 2 P 4 O 10 Si O 2 SO 2 A . Acidic 10 Amphoteric 2 Alkaline 4 B . Acidic 7 Amphoteric 1 Alkaline 5 C. Acidic 9 Amphoteric 2 Alkaline 2 D. Acidic 6 Amphoteric 1 Alkaline 6 E. Acidic 8 Amphoteric 1 Alkaline 4
Слайд 14

Indicators of chemical reactions Emission of light or heat Formation of a gas Formation of a precipitate Color change Emission of odor
Слайд 15

Describing Chemical Reactions Atoms aren t created or destroyed. A chemical equation should be balanced. Sulfur reacts with oxygen to form/to give sulfur dioxide. One mole of sulfur reacts with one mole of oxygen forming/giving one mole of sulfur dioxide. Sulfur, a yellow solid, burns forming a colorless gas with an irritating smell. sulfur oxygen sulfur dioxide S (s) O 2(g) SO 2(g)
Слайд 16

1. Synthesis Reactions
Реакция соединения occurs when two or more simple substances combine to produce a more complex substance. AKA: C ombination reaction . A B AB HINT : only one product. Examples of Synthesis Reactions CO 2 H 2 O H 2 CO 3 4Fe 3O 2 2Fe 2 O 3 Li 2 O H 2 O 2Li OH
Слайд 17

2. Decomposition Reactions
Реакция разложения occurs when a complex substance is broken down into two or more simpler substances. Heat is often used to aid in decomposition reactions – these reactions that employ heat are called thermal decompositions . Decompositions and synthesis reactions are opposites. AB A B HINT : only one reactant, two or more products. Examples of Decomposition Reactions: NH 4 NO 3 N 2 O 2H 2 O Ca(OH) 2 Ca O H 2 O 2H 2 O 2 2H 2 O O 2
Слайд 18

3. Single Displacements
Реакция замещения occurs when a single element takes the place of one of the elements in a compound. AKA: Single Replacement AB Z ZB A Metals displace metals while nonmetals displace nonmetals. HINT: The single mysterious loner moves into town and breaks up the happy couple! Examples of Single Displacement Reactions Fe Cu SO 4 Fe SO 4 Cu 2K Mg O K 2 O Mg 2Cu F Ba Ba F 2 2Cu
Слайд 19

Using the Activity Series The activity series (электрохимический ряд напряжений металлов) is a list of metals and hydrogen that are arranged in order of reactivity. Li K Ba Ca Na Mg Al Zn Fe Ni Sn Pb H Cu Hg Ag Au The rule is that the element can only be displaced by another element that is to the left of it. This makes Lithium the strongest and Gold the weakest. There is also a halogen activity series – it is used to predict reactions with halides. F Cl Br I
Слайд 20

Using the Activity Series You can use the activity series in three ways: Straight forward Single Displacements Reactions with Acids Reactions with Water Straight Forward Single Displacements Use the rule of whoever is more to the left wins to see if there is a reaction or not. Reactions with Acids Acids contain hydrogen (positive like the metals). If you are to the left of hydrogen – you react and take its place – if you are to the right – there is no reaction. Reactions with Water Only the first five elements (Li K Ba Ca Na) will react with water. It will form a hydroxide and hydrogen gas.
Слайд 21

4. Double Displacements
Реакция обмена always involves two ionic compounds that switch partners with each other. Again, positive ions switch with positive ions (and/or vice-versa). AB XY AY XB HINT : Two couples switch partners at the dance. Examples of Double Displacement Reactions: Pb(NO 3 ) 2 2KI Pb I 2 2KNO 3 Na 2 SO 3 2HCl 2Na Cl H 2 SO 3 2Na OH H 2 SO 4 2H 2 O Na 2 SO 4
Слайд 22

Indicators of occurring reactions Precipitate – solid formed from two liquids. Gas – some compounds form products that break down further into gases. Water – results from a neutralization between an acid and a base. Pb(NO 3 ) 2 (aq) K 2 Cr O 4 (aq) Pb Cr O 4 (s) 2KNO 3 (aq)
Слайд 23

5. Combustion Reaction
Реакция горения occurs when a substance (the fuel ) reacts very rapidly with oxygen to form carbon dioxide and water. Combustion reactions release a good deal of energy in a very short period of time. Fuel O 2 CO 2 H 2 O HINT : Something combines with oxygen to produce carbon dioxide and water. C 10 H 8 (s) 12O 2 (g) 10CO 2 (g) 4H 2 O(g )
Слайд 24

Incomplete Combustion
If a combustion occurs at a lower temperature, it may result in an incomplete combustion. The products of an incomplete combustion are water, carbon dioxide, carbon monoxide and carbon (a solid residue). The general equation is: Fuel O 2 H 2 O CO 2 CO C
Слайд 25
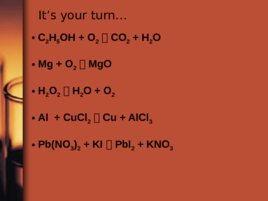
It s your turn C 2 H 5 OH O 2 CO 2 H 2 O Mg O 2 Mg O H 2 O 2 H 2 O O 2 Al Cu Cl 2 Cu Al Cl 3 Pb(NO 3 ) 2 KI Pb I 2 KNO 3
Слайд 26

Oxides Compounds of oxygen with other elements are called oxides. NO 2 , SO 2 , H 2 O, CO 2 , N 2 O 5 , NO, N 2 O are common non metal oxides, they have covalent bond structure. Na 2 O, Fe O, Al 2 O3, Ca O, Si O 2 , Mg O, Cu O, Pb O are some common metal oxides they have ionic structure. Naming of Oxides They are named like binary compounds. Mg O : Magnesium oxide SO 2 : Sulfur dioxide P 2 O 5 : Diphosphorus pentoxide Sn O 2 : Tin(IV) oxide
Слайд 27

Classification of Oxides
1. Acidic Oxides Oxygen rich compounds of non metals are called acidic oxides. SO 2 , NO 2 , P 2 O 5 , Cl 2 O are examples. Their solutions are acidic. They are known as acidic anhydrides. Acidic oxide water Acid P 2 O 5 3H 2 O 2H 3 PO 4 2. Basic Oxides Generally metal oxides are called basic oxides. Na 2 O, Ca O, Mg O are examples. Their solutions are basic. They are known as basic anhydrides. Basic oxide water Base Mg O H 2 O Mg(OH) 2 3. Mixed Oxides Compounds that contain two oxides of the same metal are called mixed oxides. Fe 3 O 4 , Mn 3 O 4 , Pb 3 O 4 are examples. They behave as if they are two separate oxides in chemical reactions. Fe 3 O 4 : Fe O Fe 2 O 3 : Iron (II, III) oxide
Слайд 28

Bases Compounds dissolving in water by producing OH - ion are called bases. They have slippery feeling. Many cleaning products contain bases. Na OH(s) Na (aq) OH - (aq) Naming of Bases The word hydroxide is added after the name of metal ion in the naming of bases. Mg(OH) 2 : Magnesium hydroxide KOH : Potassium hydroxide Na OH : Sodium hydroxide Ba(OH) 2 : Barium hydroxide
Слайд 29

Classification of Bases
According to Strength Bases that ionize in water completely are said to be strong base. Na OH, KOH and Li OH are strong bases (alkalis). Bases that ionize in water partially are called weak bases. Fe(OH) 2 , Al(OH) 3 are example for weak bases. Chemical Properties of Bases According to solubility bases conduct electricity. change the color of litmus paper to blue. react with acids and produce salt and water. Water insoluble bases decompose on heating to give metal oxides and water. 2KOH(s) H 2 SO 4 ( l ) K 2 SO 4 (aq) 2H 2 O( l ) Mg(OH) 2 Mg O H 2 O
Слайд 30

Acids Compounds dissolving in water by producing H ion are called acids. HCl(g) H (aq) Cl - (aq) H 2 SO 4 2H (aq) SO 4 -2 (aq) They have sour taste. They change the color of litmus paper to red. Their aqueous solutions conduct electricity. They are corrosive substances. Most of them are soluble in water. Naming of Acids Acids containing two types of atoms are called binary acids. Their names follow the form hydro nonmetal name –ic acid . HCl : Hydrochloric acid Acids containing oxygen atoms are called oxy acids. Their names follow the form –ic acid, or –ous acid. HNO 3 : Nitric acid HNO 2 : Nitrous acid
Слайд 31

Classification of Acids According to Strength If an acid ionizes completely, it is an strong acid, and if it ionizes partially it is a weak acid. Strong acids HCl, H2SO4, HNO3 Weak acids H2SO3, HNO2, H2S, HCN According to Number of Hydrogen Atoms According to number of H ion produced acids are classified as monoprotic, diprotic or triprotic. Monoprotic acids HCl, HNO3, HI, HBr, HCl O4 Diprotic acids H2SO3, H2S, H2CO3, H2SO4 Triprotic acids H3PO4
Слайд 32

Chemical Properties of Acids Acids ionize in water and conduct electricity, during the ionization heat is released. They change the color of indicators. They react with bases and produce salt and water, it is called neutralization reaction. They react with basic oxides and some salts. They react with some metals and produce hydrogen gas. HNO 3 ( l ) KOH(s) KNO 3 (aq) H 2 O( l ) 2HCl(aq) Zn(s) Zn Cl 2 (aq) H 2 (g)
Слайд 33

Amphoteric Compounds Most of the compounds of Zn, Al, Cr, Sn, Pb, and Be are amphoteric compounds. Oxides and hydroxides of these metals have both acidic and basic characters. They are insoluble in water and do not react with it. Zn O, Al 2 O 3 are oxides, and Zn(OH) 2 , Al(OH) 3 are hydroxides. Zn O 2HCl Zn Cl 2 H 2 O Zn O 2Na OH Na 2 Zn O 2 2H 2 O
Слайд 34

p H p H is a numeric scale used to specify the acidity or basicity of an aqueous solution. It is approximately the negative of the logarithm to base 10 of the molar concentration, measured in units of moles per liter, of hydrogen ions. More precisely it is the negative of the logarithm to base 10 of the activity of the hydrogen ion. p H - lg H
Слайд 35

Salts Salts are ionic compounds of anions and cations: Na Cl, Ca CO 3 , Zn Br 2 , Fe SO 4 etc They are all crystalline solids. They have high melting and boiling points. Many of them are soluble in water and their aqueous solutions conduct electricity. Naming of Salts In the naming of salts first metal ion (positive ion) then name of negative ion is read. KMn O 4 Potassium permanganate
Слайд 36

Classification of Salts A. Neutral Salts are formed from the reactions of strong acids with strong bases. Na Cl, Li NO 3 , KNO 3 , Li 2 SO 4 . B. Acidic Salts are formed from the reactions of strong acids with weak bases. Their solutions are acidic. Fe Cl 2 , Zn(NO 3 ) 2 C. Basic Salts are formed from the reactions of weak acids with strong bases. Their solutions are basic. Na CN, Li F, K 2 CO 3 , K 2 C 2 O 4
Слайд 37

Chemical Properties of Salts Salts can react with metals according to activity strength. Zn(s) 2Ag NO 3 (aq) 2Ag(s) Zn(NO 3 ) 2 (aq) Water soluble salts undergo displacement reaction. KCI(aq) Ag NO 3 (aq) 2Ag Cl(s) KNO 3 (aq) They may also react with acids under certain conditions. 2HCI Ca CO 3 Ca Cl 2 H 2 O CO 2
Слайд 38

The end
^ Наверх
X
Благодарим за оценку!
Мы будем признательны, если Вы так же поделитесь этой презентацией со своими друзьями и подписчиками.